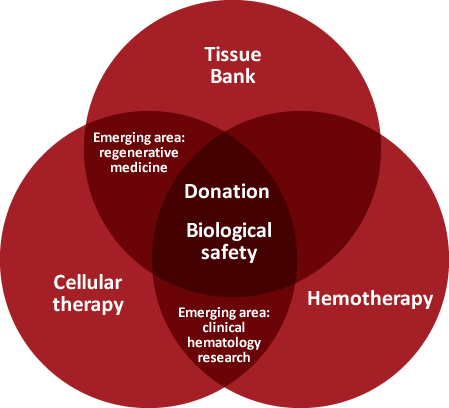
The Banc de Sang i Teixits (Blood and Tissue Bank, BST) regards research as a strategic activity for providing services of the highest quality and safety, rapidly incorporating improvements in our field as they emerge, and working to devise new diagnostic and therapeutic tools.
Research
We conduct independent research projects and work on others in cooperation with every centre pertaining to the Catalan Health Institute, with the majority of centres in the Network of Public-Access Hospitals and with Catalan universities, and we also pursue strategic alliances with research centres around the world.
Download the Scientific Research Report 2022
At present, we are involved in 60 research and innovation projects at the BST. Within our organisation, 55 people dedicate part of their time to research.
Funded projects
- Lab-grown red cells for the transfusion management of vaso-occlusive crisis in sickle cell patients" amb expedient HR23-00367 està finançat pel Programa CaixaResearch Health de la Fundació "la Caixa HR23-00367, funded by Programa CaixaResearch Health - Fundació "la Caixa"

- VASCRAFT: New human decellularized and re-endothelialized tissue-engineered VASCular gRAFT for coronary artery bypass grafting. File: CPP2021-008438, funded by the call of RDi Projects in Public-Private Collaboration of the National Program to enhance the Scientific-Technic Research and its Transference, of the National Plan of Scientific, Technic and Innovation Research 2021-2023, within the framework of the Recovery, Transformation and Resilience Plan. Funded by:

- OSCAR-iNK: Off-the-Shelf iPSC-derived next generation CAR-NK cells for solid tumor allogenic immunotherapy. File: CPP2021-008350, funded by the call of RDi Projects in Public-Private Collaboration of the National Program to enhance the Scientific-Technic Research and its Transference, of the National Plan of Scientific, Technic and Innovation Research 2021-2023, within the framework of the Recovery, Transformation and Resilience Plan. Funded by:

- The project "UNiKAR: Universal cord blood iPSC-derived CAR-NK cells for an 'off-the-shelf' immunotherapy for cancer" is funded by Carlos III Health Institute and cofunded by the European Union by FEDER.

- The project "HEMO-GAS. RECREATING THE EMBRYONIC NICHE FOR HEMATOPOIETIC STEM CELL PRODUCTION AND DERIVATIVES IN HUMAN GASTRULOIDS. PLEC2021-007518", is funded by the Science and Innovation Ministry/State Research Agency (Digital Object Identifier 10.13039/501100011033) and by the European Union "NextGenerationEU"/PRTR. Project PLEC2021-007518 funded by:

-
Development of a dual CD1a/CCR9-directed CAR-T therapy for the treatment of R/R T-cell Acute Lymphoblastic Leukemia. File: CPP2022-009759, funded by MCIN/AEI /10.13039/501100011033 and European Union NextGenerationEU/ PRTR.

-
EPIFIN: Engineering induced Pluripotent stem cells for a novel Immunotherapy of Fungal INfections. File: CPP2022-009594, funded by MCIN/AEI /10.13039/501100011033 and European Union NextGenerationEU/ PRTR.

-
LAB-REDCELL: Lab-grown red cells from immortalized erythroid cell lines as an alternative source of red cells with rare phenotypes for diagnostic and therapeutic applications. File: PID2022-141849OB-I00 funded by MCIN /AEI /10.13039/501100011033 / and by FEDER, A way to make Europe.

-
RELECOV 2.0 Consolidation of WGS and RT-PCR activities for SARS-CoV-2 in Spain towards sustainable use and integration of enhanced infrastructure and processes in the RELECOV network. Project co-funded by European Union

Scientific output
By 2022 BST researchers published 54 articles in scientific journals that had an impact factor of 488. 51% of these publications are from the first quartile.
Patents
BST has one patent granted in Spain, one in USA and two granted by the European Patent Office validated in several European countries.